 🛡️ Public Health
🛡️ Public Health
India • February 28, 2026
India Launches Nationwide Single-Dose HPV Vaccination Drive for Girls Aged 9–14
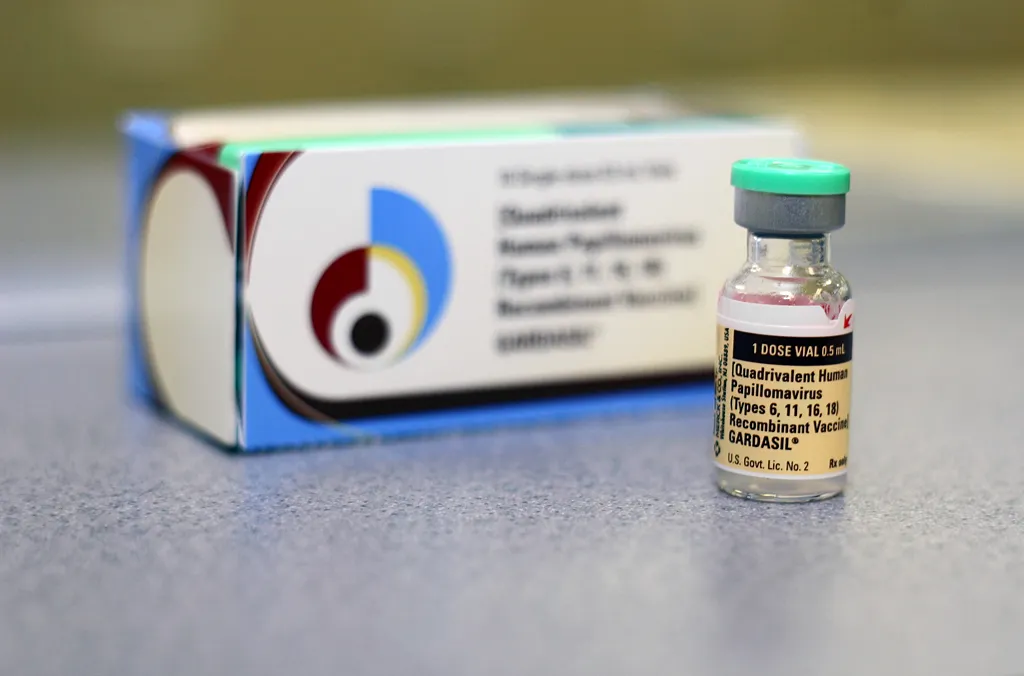
Prime Minister Modi launches the campaign from Ajmer, Rajasthan, targeting girls aged 9–14 years with a free, voluntary single dose. The programme leverages the U-WIN digital platform and includes both Gardasil-4 and the indigenous Cervavac vaccine.
India has officially rolled out its nationwide Human Papillomavirus (HPV) vaccination programme, targeting girls aged 9–14 with a single dose. Prime Minister Narendra Modi launched the campaign on February 28, 2026, from Ajmer, Rajasthan [citation:6][citation:10]. This free and voluntary drive aims to prevent cervical cancer, which remains the second-most common cancer among women in India. The programme utilizes both Gardasil 4 (MSD) and the indigenous Cervavac (Serum Institute of India) following recommendations from the National Technical Advisory Group on Immunisation (NTAGI) [citation:2][citation:3]. With an estimated 80,000 new cases and 35,000–42,000 deaths annually, this initiative represents a critical step toward eliminating a largely preventable disease [citation:1][citation:2].
What the Vaccine Protects Against
Tap a type to see what it causes and why it matters.
Everything You Need to Know
The drive targets girls aged 9 to 14 years, with those turning 15 during the 90-day campaign also eligible [citation:10]. The vaccine is free of cost and voluntary, administered at government facilities including Ayushman Arogya Mandirs, District Hospitals, and Government Medical Colleges. Parental or guardian consent is mandatory before vaccination, following an “opt-in” requirement via the U-WIN platform or physical form [citation:10]. The special campaign runs over a three-month window, with doses available daily at participating facilities. States like Himachal Pradesh have scheduled specific session days (e.g., March 29, April 5) to cover eligible girls [citation:5]. More on India’s regulatory processes for public health.
India has adopted a single-dose primary schedule, backed by a 2022 WHO position paper which confirmed single-dose protection is comparable to multi-dose regimens for individuals aged 9–20 years. Studies show HPV vaccines are 93–100% effective in preventing cervical cancer caused by covered HPV types when administered before viral exposure [citation:1]. An optional delayed second dose (3–5 years later) remains available under the national plan. Over 37 countries have adopted single-dose schedules, and globally, more than 500 million doses have been administered since 2006, per Gavi data [citation:3].
The national programme is utilizing both Gardasil 4 (quadrivalent), manufactured by MSD (Merck), and the indigenously developed Cervavac from the Serum Institute of India [citation:2][citation:3]. Both vaccines protect against HPV types 6, 11, 16, and 18. Cervavac has been incorporated as a key pillar of the strategy under the “Atmanirbhar Bharat” initiative and is licensed for use in India. Vaccine procurement is supported through government tenders and global supply arrangements. In the private market, Gardasil 9 (nine-valent) and other options remain available at varying costs. India now joins over 160 countries with HPV vaccination in their national programmes [citation:7].
All vaccination sessions are staffed by trained healthcare teams for post-vaccination observation and immediate AEFI (Adverse Events Following Immunisation) management. Vaccination centres are linked to 24/7 government health facilities and maintain Cold Chain Points (2-8°C) for vaccine integrity [citation:10]. An ICMR inquiry into the 2009–10 trials (Andhra Pradesh and Gujarat) concluded that the deaths among trial participants were “most probably unrelated to the vaccine”, while recommending strengthened AEFI investigation and community engagement for future drives. The current programme includes structured AEFI reporting pathways and anaphylaxis management kits at all sites [citation:10]. See also: how environmental health factors compound disease risk.
Vaccination alone does not eliminate the need for cervical cancer screening. India’s current national screening coverage stands at a critically low 1.9%–2.2% of women aged 30–49, according to NFHS-5 and NNMS data [citation:4][citation:8]. The HPV drive will be complemented by calls to expand screening programmes — evidence-based linkage between vaccination, screening and timely treatment is the complete prevention pathway. The WHO Cervical Cancer Elimination Initiative sets the 90-70-90 targets by 2030. Advances in medical research continue to broaden the tools available for long-term health management.
The national programme uses a single-dose schedule based on a 2022 WHO position paper confirming comparable protection from one dose for those aged 9–20 years. An optional delayed second dose (3–5 years later) is available in the national plan. A single-dose strategy maximises coverage, simplifies cold-chain logistics and reduces the risk of drop-off between doses — critical factors for a campaign targeting millions of girls annually. This approach follows successful single-dose rollout evidence from multiple national programmes globally.
In private medical settings, HPV vaccines are typically administered in two or three doses over several months depending on age and clinical guidance. Gardasil 9 (nine-valent), Gardasil 4, and Cervavac are available privately. Multi-dose schedules were the standard before single-dose evidence from the WHO and multiple national studies confirmed that one dose provides robust protection in the eligible age group for the national programme’s target cohort.
India’s HPV Vaccine Journey
On-Ground Safety Protocols
- ✓Your daughter should be aged 9 to 14 years to be eligible. Those turning 15 during the 90-day campaign can also receive the shot [citation:10].
- ✓The vaccine is completely free of cost at all government facilities — Ayushman Arogya Mandirs, District Hospitals and Government Medical Colleges.
- ✓Your written or digital consent is mandatory before vaccination. No vaccine is given without it [citation:10].
- ✓The vaccine is non-live and recombinant — it cannot cause HPV infection. It triggers an immune response only.
- ✓Plan for a 30-minute on-site observation window post-vaccination for safety monitoring [citation:10].
- ✓A QR-coded digital certificate will be issued via the U-WIN platform — keep it safe as a permanent health record.
- ✓Vaccination does not replace cervical cancer screening. Discuss regular screening schedules with a healthcare provider as your daughter grows older.
- ✓Former ICMR Director General V.M. Katoch described the nationwide rollout as a “landmark moment in India’s public health journey” and a “visionary and transformative step” [citation:7].
The nationwide HPV vaccination programme was launched by Prime Minister Modi in Ajmer on February 28, 2026, targeting girls aged 9–14 with a free, voluntary single dose. The campaign utilizes both Gardasil 4 and the indigenous Cervavac vaccine, with all doses tracked via the U-WIN digital platform. Safety protocols include mandatory parental consent, trained health workers, cold chain maintenance, and AEFI management systems. The programme addresses India’s high burden of cervical cancer while acknowledging the need to expand screening coverage from its current low levels. Read more about advances in preventive medical technology and India’s broader public health challenges.