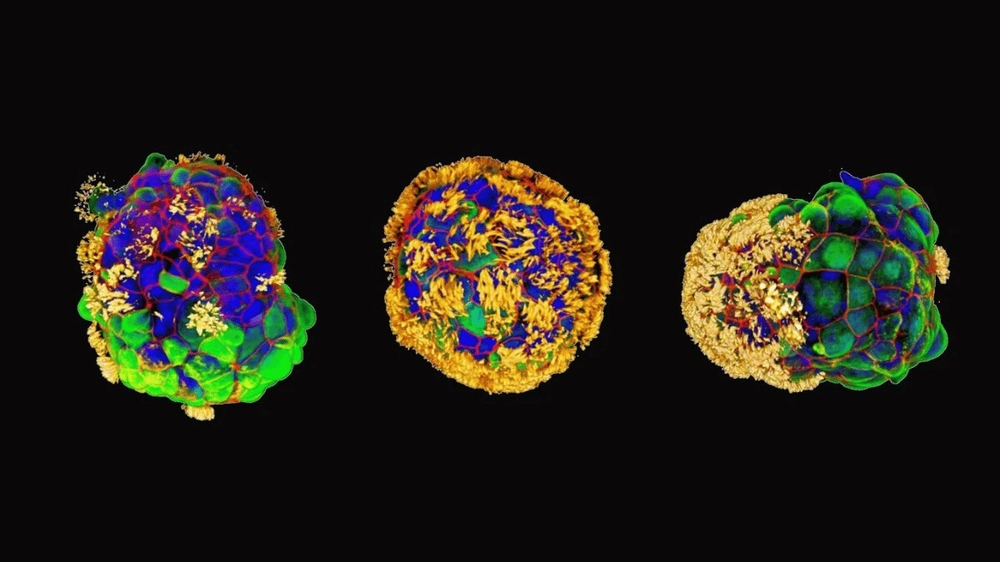
Tufts University researchers have engineered “Anthrobots” from human tracheal cells. These tiny, cell-made robots, as small as a hair’s width or as large as a pencil point, are a blend of biology and robotics. They’re designed to self-assemble and have shown impressive healing effects on other cells.
The big deal here is that these Anthrobots are made from adult human cells – no genetic modifications! They come from the trachea, the air passage to our lungs, and are equipped with tiny hair-like structures called cilia. These cilia are the heroes, driving the Anthrobots’ movements across surfaces.
Now, let’s talk size and shape. These bots vary from 30 to 500 micrometers. Their movements? Quite diverse – some move in straight lines, others in circles, or combine both movements. There’s even a group that just wiggles around. The team, including Ph.D. student Gizem Gumuskaya, observed different shapes and movement types, revealing a fascinating aspect of this biorobotics platform.
We wanted to probe what cells can do besides create default features in the body. By reprogramming interactions between cells, new multicellular structures can be created, analogous to the way stone and brick can be arranged into different structural elements like walls, archways or columns.
Gizem Gumuskaya, Ph.D. student
But why are they special? These Anthrobots have demonstrated their capability to move across human neurons and encourage growth, especially in regions with cell damage. This is huge for regenerative medicine. For example, the Anthrobots can cross and repair gaps in human neuronal layers.
Gumuskaya, with an architecture background, likens the process to arranging bricks and stones into various structures. The idea is to reprogram cell interactions to create new multicellular structures.
The cellular assemblies we construct in the lab can have capabilities that go beyond what they do in the body. It is fascinating and completely unexpected that normal patient tracheal cells, without modifying their DNA, can move on their own and encourage neuron growth across a region of damage.We’re now looking at how the healing mechanism works, and asking what else these constructs can do.
Michael Levin, Vannevar Bush Professor of Biology and Josh Bongard at the University of Vermont
Similar Posts
The method? Start with a single tracheal cell. In the lab, these cells naturally form multicellular spheres called organoids. By tweaking the growth conditions, researchers made the cilia face outward, which led to the organoids’ movement.
But here’s where it gets even more interesting. The researchers created “superbots” – clustered assemblies of Anthrobots. They placed these superbots near a lab-created ‘wound’ in a layer of human neurons. Without any genetic modification, these Anthrobots triggered significant neuron growth, bridging the gap in the damaged area.
Levin, one of the lead researchers, notes the potential for Anthrobots to travel through the body, respond to their environment, or help build engineered tissues in the lab. The possibilities are vast – from clearing arterial plaque to repairing nerve damage or delivering targeted drugs.
One key advantage of using human cells is that Anthrobots built from a patient’s own cells won’t trigger immune responses. Plus, they biodegrade naturally in about 45-60 days. They’re safe, with no risk of unintended spread outside the lab, as they can only survive under specific lab conditions and don’t reproduce.
Summing it up, Anthrobots are a blend of bioengineering and robotics, representing a fresh approach in regenerative medicine and beyond. They’re a testament to the untapped potential in human cells and the innovative ways we can use them.