A breakthrough study from MIT engineers reveals that cells can “remember” gene activity at various intensity levels, challenging the long-held belief that cellular memory operates as a simple on/off switch.
This discovery could reshape our understanding of how cells maintain their identity and potentially lead to new approaches in tissue engineering and cancer treatment. Published September 9, 2025, in Cell Genomics, the research shows that cells can lock genes at intermediate expression levels—not just fully activated or fully repressed—and maintain these levels for extended periods.
“Our finding opens the possibility that cells commit to their final identity by locking genes at specific levels of gene expression instead of just on and off,” explains Domitilla Del Vecchio, professor of mechanical and biological engineering at MIT and study author. “The consequence is that there may be many more cell types in our body than we know and recognize today, that may have important functions and could underlie healthy or diseased states.”
For decades, scientists believed that epigenetic memory—the process by which cells retain gene expression patterns that define their identity—worked in binary fashion. This binary model suggested that genes were either completely activated or completely turned off, much like a light switch.
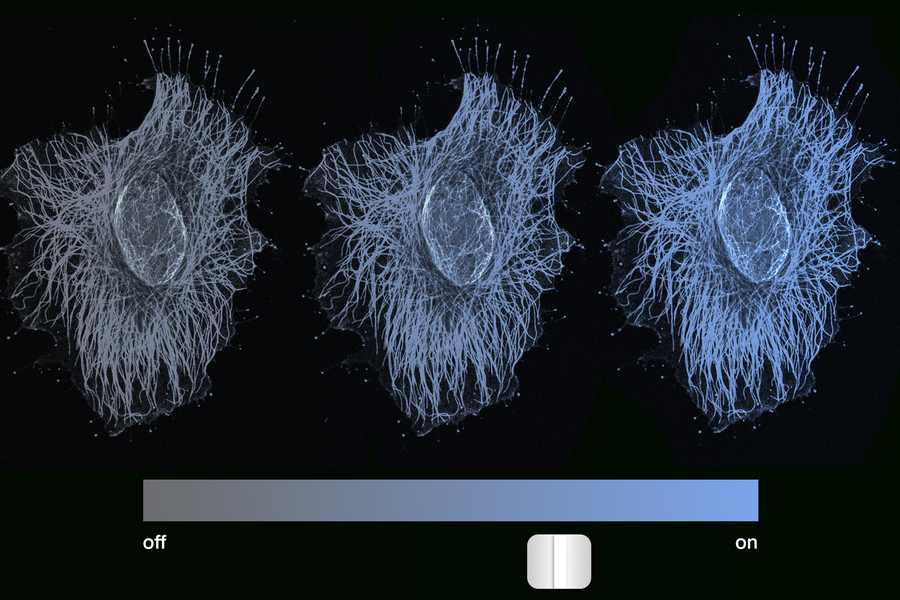
To test this assumption, the MIT team conducted experiments using hamster ovarian cells. They engineered a single gene to express at different levels in different cells and paired it with a blue fluorescent marker that glowed with brightness corresponding to the gene’s expression level.
After briefly triggering DNA methylation—a natural mechanism that helps lock gene expression in place—researchers tracked the cells for five months. What they discovered was surprising: cells maintained their original expression levels across the entire spectrum, from fully active to completely inactive, with every gradation in between.
Similar Posts
“Our fluorescent marker is blue, and we see cells glow across the entire spectrum, from really shiny blue, to dimmer and dimmer, to no blue at all,” Del Vecchio notes. “Every intensity level is maintained over time, which means gene expression is graded, or analog, and not binary.”
This analog memory capability could explain why certain cancer cells develop resistance to therapies. It might also open new possibilities for creating more sophisticated artificial tissues by fine-tuning gene expression like adjusting a radio dial rather than simply flipping switches.
Michael Elowitz, professor of biology and biological engineering at the California Institute of Technology, who was not involved in the study, comments: “Del Vecchio and colleagues have beautifully shown how analog memory arises through chemical modifications to the DNA itself. As a result, we can now imagine repurposing this natural analog memory mechanism, invented by evolution, in the field of synthetic biology, where it could help allow us to program permanent and precise multicellular behaviors.”
Lead author Sebastian Palacios emphasizes the significance of this finding: “One of the things that enables the complexity in humans is epigenetic memory. And we find that it is not what we thought. For me, that’s actually mind-blowing. And I think we’re going to find that this analog memory is relevant for many different processes across biology.”
While the current research focused on a single engineered gene in hamster ovarian cells, future studies will need to determine how widespread this analog memory is across different genes and cell types, particularly in human cells.
The research received support from the National Science Foundation, MODULUS, and a Vannevar Bush Faculty Fellowship through the U.S. Office of Naval Research.